A Data-Based Comparison of Wavefront Lasers
Any intelligent consumer seeking information about laser vision correction these days is assaulted and often overwhelmed by slick marketing and confusing representations about the different lasers used for treatment. Particularly in the arena of wavefront lasers, the information and hype is bewildering. At LA Sight, we don’t think it needs to be this way.
The good news is that that all modern wavefront lasers are technically superior to their non-wavefront-capable predecessors, and create optical results that are slightly to significantly better for patients. But it’s still confusing to wade through all the information about “wavefront-customized” and “wavefront-optimized” treatments, to try and discern the nuances and advantages of each. Fortunately, it is possible now to compare these different lasers by carefully looking at the data presented to the FDA as a part of the regulatory approval process. That’s exactly what we present here. Of course, it helps to be a professional and expert in the field, so we chose to reprint an article originally intended for LASIK surgeons, by a recognized authority with experience in both surgical outcomes analysis and the FDA clinical trial submission process. Comments and clarifications by Dr. Wallace are denoted by [brackets].
A Closer Look at the Options for LASIK Surgery
Guy M. Kezirian, MD, FACS Reprinted with permission from Review of Ophthalmology, November ’04
The 1999 announcement by Theo Seiler, MD, PhD, that he had performed LASIK based on aberrometry data rather than a refraction set off a frenzy of research and development.
For the refractive surgeon, the target of the wavefront marketing that also exploded around that same time, the situation can be confusing. Should I upgrade to wavefront? Will the marketplace support a new capital investment? Will I be left behind if I don’t join in? Are wavefront-customized treatments better than wavefront-optimized treatments?
These issues raise the question: Which LASIK is best for my patients? To determine the answer, we must look at data from well-conducted studies.
Fortunately, data are readily available from the recent Food and Drug Administration trials that led to two aberrometer-based wavefront approvals (LadarVision and the Visx S4) and from WaveLight’s FDA approval of the Allegretto Wave laser with wavefront-optimized profiles. Differences in study protocols make true head-to-head comparisons of the systems impossible, and multiple components contribute to visual quality. I encourage surgeons to seek all available data when considering the options.
Background
Until recently, refractive surgery has been based on clinical refractions. Corrections were limited to sphere and cylinder with no consideration to optical irregularities such as coma, spherical aberration or other aberrations. Yet non-spherocylindrical aberrations do exist in some eyes, and visual function may be improved if they are treated.
 Aberrometers measure all the optical errors of the eye—sphere, cylinder and aberrations such as coma, spherical aberration, and others. Non-spherocylinder aberrations are referred to as higher-order aberrations (HOA) to distinguish them from spherocylinder errors, or lower-order aberrations.
Aberrometers measure all the optical errors of the eye—sphere, cylinder and aberrations such as coma, spherical aberration, and others. Non-spherocylinder aberrations are referred to as higher-order aberrations (HOA) to distinguish them from spherocylinder errors, or lower-order aberrations.
Aberrometer technologies are new and evolving, as is the science used to transform aberrometer measurements into excimer laser ablation patterns. Aberrometer-based treatments to treat HOAs in addition to the spherocylindrical error are being marketed as “customized” treatments.
Originally, it was hoped that wavefront technologies would eliminate postop HOAs and provide patients with “super-vision.” This has proved to be an elusive goal, [that’s a nice way of saying, “it hasn’t worked out”] and postoperative corneas may still have more HOAs than they started with. The main culprit is spherical aberration, which early lasers induced in large amounts, and which newer lasers are trying to prevent.
Prof. Seiler’s first aberrometer-guided treatments spurred laser manufacturers to develop wavefront systems. WaveLight, the manufacturer of the wavefront system that Prof. Seiler originally used for the first aberrometer-guided treatments, de-emphasized aberrometer-based ablations for every eye. Instead, they applied wavefront data to modify the conventional treatments in the Allegretto Wave laser. WaveLight calls the new profiles “wavefront-optimized” because they are based on Prof. Seiler’s original data from aberrometer-based, wavefront treatments.
The main difference is that these profiles have been shown to induce less spherical aberration than conventional treatments. They do so by adding more spots in the periphery in an attempt to preserve the original asphericity of the eye. Wavefront-optimized ablations do not correct other pre-existing aberrations, however, and operate under the assumption that preventing the spherical aberration that is induced by standard LASIK is more important than correcting the small aberrations such as coma that are present in some eyes.
 These treatments do not require aberrometry measurements; they are based on the spherocylinder refraction of the eye. The other companies—Visx, Bausch & Lomb, and Alcon—all attempt to correct pre-existing aberrations and provide “custom” treatments for each eye, based on per-eye aberrometry measurements.
These treatments do not require aberrometry measurements; they are based on the spherocylinder refraction of the eye. The other companies—Visx, Bausch & Lomb, and Alcon—all attempt to correct pre-existing aberrations and provide “custom” treatments for each eye, based on per-eye aberrometry measurements.
To date, Alcon’s LadarVision CustomCornea, Visx’s CustomVue, WaveLight’s Allegretto Wave laser and, just last month, Bausch & Lomb’s Zyoptics have been approved in the United States. As of this writing, the FDA had not yet released the B&L data, so it is not included in this analysis.
About the Lasers
The Visx S4 laser is a time-tested platform with a track record for reliable performance. It is based on a broad-beam platform and has recently incorporated an active tracker and a variable spot scanning system to allow the spot size to change from 0.65 to 6 mm during treatments. Astigmatism is corrected by reducing the diameter of the ablation zone in one meridian, resulting in more correction along the shorter diameter. The Visx CustomVue approval is for treatments of up to -6 D spheroequivalent with up to 3 D astigmatism. The optical zone diameter is 6 mm.
Alcon purchased the Autonomous laser from Summit Technologies Inc. and modified it to become the LadarVision 4000 laser. The laser is a flying spot laser with an active tracking device. Its CustomCornea approval is for spherical treatments up to -7 D of myopia with less than 0.5 D of astigmatism, and it uses a 6.5 mm optical zone.
The Bausch & Lomb Technolas 217z customized ablation system was approved to correct up to -7 D of myopia with up to -3 D of astigmatism.
The Allegretto Wave was developed specifically for wavefront treatments—with a fast, accurate tracker, a small 0.95-mm spot size, a rapid 200-Hz repetition rate, and a Gaussian beam profile that is particularly well-suited to treating localized aberrations. The Allegretto Wave U.S. clinical trial utilized the wavefront-optimized profiles. Further, prior to beginning the trials, the laser algorithm was adjusted to minimize the need for nomogram adjustments. This was done using the approach that Houston surgeon Jack T. Holladay, MD, MSEE, and I developed for the Refractive Surgery Consultant Elite software.
Together, these three factors—new technology, wavefront-optimized profiles, and a nomogram-adjusted internal algorithm—led to the Allegretto Wave results shown below. As you will see, the results are better than any reported to date using custom wavefront in most categories.
Methodology
FDA study results were used to compare the outcomes of three wavefront-based systems. Since the studies were reported using slightly different categories and treatment ranges, two sets of comparisons are made.
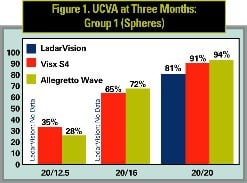
 The first set compares results in eyes operated on for spherical corrections for Visx S4, and for up to 0.5 D of cylinder for LadarVision and the Allegretto Wave. Sphere treatments were reported for all three lasers so this comparison provides a good overview.
The first set compares results in eyes operated on for spherical corrections for Visx S4, and for up to 0.5 D of cylinder for LadarVision and the Allegretto Wave. Sphere treatments were reported for all three lasers so this comparison provides a good overview.
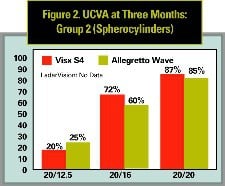
Results for spherocylinder treatments are not available with the LadarVision, but they are available for the Visx S4 and Allegretto Wave lasers, so a second comparison set (See Figure 2) is provided to compare spherocylinder outcomes. For all comparisons, only single-procedure results are reported—no reoperation results are included, and all eyes were targeted for distance vision.
All three studies reported data through one year, however, reported follow-up in the Visx S4 study falls off rapidly after three months. Of the 351 Visx S4 eyes operated, 318 (90.5 percent) were reported at three months, but only 86 (24.5 percent) were reported at one year. To allow adequate representation of the Visx S4 results, the three-month interval from all three studies was used for comparison here.
 The Visx S4 study only reported treatments up to 6 D spheroequivalent. Alcon’s LadarVision study reported up to 7 D but only 4/139 eyes (2.8 percent) had more than 6 D treated. Therefore, to keep the groups similar, results through 6 D for each laser are compared here; the four LadarVision eyes over 6 D are excluded.
The Visx S4 study only reported treatments up to 6 D spheroequivalent. Alcon’s LadarVision study reported up to 7 D but only 4/139 eyes (2.8 percent) had more than 6 D treated. Therefore, to keep the groups similar, results through 6 D for each laser are compared here; the four LadarVision eyes over 6 D are excluded.
A final note: Alcon reported two groups of data in its results—a “Safety Cohort,” 426 eyes operated with several protocols, and an “Effectiveness Cohort” of the 139 eyes that were used to report results such as UCVA and refractive outcomes in order to gain FDA approval. The Effectiveness Cohort is used here, except as noted.
Factors Limiting Comparison
This comparison was performed using results from prospective, similarly designed studies under FDA protocol. Some differences in the studies exist, however, and merit consideration when interpreting the results.
The Visx S4 study prescreened eyes to require preop BSCVA of 20/20 + 3 (basically, 20/16) or better using the “PreVue” lens to enroll in the study. Alcon’s LadarVision study used 20/50 as the cut-off, and the Allegretto Wave study used 20/40. This tends to bias the outcomes in favor of Visx S4 and against the LadarVision and Allegretto Wave lasers.
Visx S4 cylinder corrections result in smaller optical zones. The method has been shown to be very accurate but the downside is that it reduces the diameter of the optical zone in one axis.
The LadarVision results reported in the FDA publications worsened over time. For example, UCVA of 20/20 or better went from 86.3 percent at one month to 80.6 percent at three months and 79.9 percent at six months. Results with the other two lasers improved over time. This may be a nomogram issue with the LadarVision, but the exact cause is uncertain.
Experience has shown that results in the field may be better than are reported in the FDA studies. This is usually the case since surgeons can make nomogram adjustments and vary their techniques more than the studies permitted. For Visx S4 and LadarVision, however, the limitations on nomogram adjustments may preclude much improvement. The LadarVision and Visx S4 FDA approvals both limit nomogram adjustments to ± 0.75 D of the sphere amount. There are no limitations on nomogram adjustments with the Allegretto Wave.
Results
Reporting compliance for all three lasers was better than 90 percent at three months, the interval used for this comparison. The refractive distribution of eyes reported at three months is similar for each laser, validating the use of the data for this comparison.
In Group 1 (spheres), there were 135 LadarVision eyes, 79 Visx S4 eyes, and 180 Allegretto Wave eyes. Group 2 (spherocylinders) included 239 Visx S4 eyes and 370 Allegretto Wave eyes. Although there were more Allegretto Wave eyes than with either of the other two lasers, and Allegretto Wave eyes tended to have higher corrections, the distribution of preoperative refractions is comparable.
Having established that the results can be compared, how do they look? There are five overriding comparisons to consider: uncorrected visual acuity; manifest refractive spheroequivalent outcomes; subjective symptoms; changes in BSCVA; and the one most important to patients—“Will I see as well after surgery without glasses as I do with my glasses before surgery?”
Uncorrected Visual Acuity
UCVA results are compared in Figures 1 and 2. Figure 1 compares Group 1 (spheres) for all lasers. Both the Visx S4 and Allegretto Wave outperform the Alcon LadarVision laser at the 20/20 level (p < 0.05). Results with the Visx S4 and Allegretto Wave at 20/16 and 20/12.5 were similar. Since the Alcon LadarVision study did not report spherocylinder results, Figure 2 compares these outcomes for the Visx S4 and Allegretto Wave lasers only. Results at the 20/20 level are statistically similar (87 percent for Visx S4 and 85 percent for the Allegretto Wave). Results at the 20/16 level are better for Visx S4 than the Allegretto Wave (72 percent v. 60 percent, p < 0.01, Chi-squared test) but slightly worse at the 20/12.5 level (20 vs. 25 percent not significant due to the small number of eyes).
Refractive Outcomes
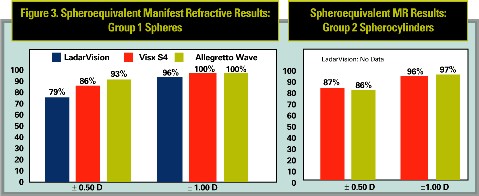
Refractive outcomes followed the same trend (See Figure 3). For spherical corrections, both Visx S4 (p = 0.03) and the Allegretto Wave (p < 0.001) outperformed the LadarVision laser at the ± 0.50 D level. The Allegretto Wave rate of ± 0.50 D was better than Visx S4 at 93 percent v. 86 percent (p = 0.08). Both the Visx S4 and Allegretto Wave had all eyes within the ± 1.00 D level, while the LadarVision rate was 96 percent (p < 0.05 and p<0.01, respectively).
Spherocylinder results for the Visx S4 and the Allegretto Wave at the ± 0.50 D and ± 1.00 D levels were very similar for both lasers.
BSCVA
The data on improved BSCVA with wavefront treatments do not appear to be as positive as early claims suggested.
BSCVA remained the same or improved in 83 percent of LadarVision eyes, 89 percent of Visx S4 eyes, and 93 percent of Allegretto Wave eyes. The differences in these rates were significantly better for the Allegretto Wave versus the other two lasers (p < 0.001 and p < 0.03, respectively) but the LadarVision and Visx S4 results were statistically equivalent (p = 0.12).
Among the LadarVision eyes, 1.4 percent eyes lost two or more lines of BSCVA. No Visx S4 eyes lost two or more lines and 0.9 percent of Allegretto Wave eyes lost two or more lines. Here, the Visx S4 results were significantly better than the LadarVision results, but not significantly different from the Allegretto Wave results. Because of the way the FDA reports were structured, it was not possible to compare the sphere and spherocylinder results separately for BSCVA.
Gain in BSCVA was more commonly seen with the Allegretto Wave (55 percent) and Visx S4 (53 percent) lasers than with the LadarVision (31 percent), though this is an example of the problem inherent in direct comparison of different studies. Due to a subtlety in the reporting format in the three studies, the Visx S4 results are reported for eyes with any gain in BSCVA—even if that gain was only one letter, and so may appear artificially better than they might if held to the same standard as the other two systems. The LadarVision and Allegretto Wave figures represent eyes that gained a full line or more of BSCVA.
These results show minimal loss of BSCVA with all three lasers. Yet, the superior results achieved with the Allegretto Wave laser suggest that aberrometry may not be the key factor in improving BSCVA.
Postop UCVA vs. Preop BSCVA
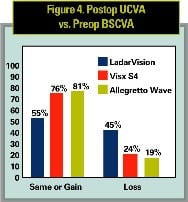
The comparison of postoperative UCVA to vision with glasses before surgery is very important to patients. The FDA now requires this measure to be published in laser labeling, so the statistic can be compared for the three lasers.
Figure 4 shows that comparison. Note that the Visx S4 study did not separate the results into sphere-only versus spherocylinder treatments, so the results for Visx S4 and the Allegretto Wave in Figure 4 include both sphere and spherocylinders. Alcon’s CustomCornea approval, it must be noted, restricted wavefront treatments to up to -7 D sphere and less than 0.50 D astigmatism.
Results show significantly better results with both Visx S4 and the Allegretto Wave than for the LadarVision laser. The Allegretto Wave results were better than Visx S4 as well, with 81 percent vs. 76 percent having the same or better UCVA after surgery than vision with glasses before surgery (p = 0.07). Only 19 percent of Allegretto Wave eyes had worse UCVA than preop BSCVA, this number was 25 percent for Visx S4 (p = 0.07) and 45 percent for LadarVision (p < 0.001 compared with Visx S4 and Allegretto Wave).
Regulatory Considerations
As noted above, the FDA approval restricts LadarVision wavefront treatments to up to -7 D sphere and less than 0.50 D astigmatism. Visx S4 wavefront treatments are approved for up to -6 D spheroequivalent with up to 3 D astigmatism. These lasers are approved for higher ranges with standard, non-wavefront treatments.
The application for FDA approval of the Allegretto Wave extended to myopia of -12 D spheroequivalent with up to 6 D astigmatism and hyperopia up to 6 D spheroequivalent and 6 D astigmatism. Wave-front-optimized ablation patterns are incorporated into all Allegretto Wave treatments.
As refractive outcome is the primary determinant of postop visual acuity, the ability to make nomogram adjustments is essential to successful implementation of wavefront LASIK. All lasers and surgeons are different and the need for nomogram adjustments for all excimer lasers is well-recognized. The Allegretto Wave permits unrestricted sphere and cylinder nomogram adjustments. The other two platforms restrict nomogram adjustments to +/- 0.75 D of the sphere.
Other Considerations
This analysis shows that the new technology, wavefront-optimized results achieved with the Allegretto Wave laser outperformed aberrometer-based LASIK with the Visx S4 and LadarVision lasers.
As new technologies and options come on line, refractive surgeons will have to consider several issues when deciding the best LASIK options for their patients.
- Cost. Custom wavefront treatments add significantly to the costs of performing LASIK. These increased costs lead patients to expect more from their treatments, and we know that unmet expectations are a significant factor in litigation. The higher costs of custom treatments is not justifiable if the aberrometers are simply acting as autorefractors, or if improvements can be traced to better lasers and better nomograms.
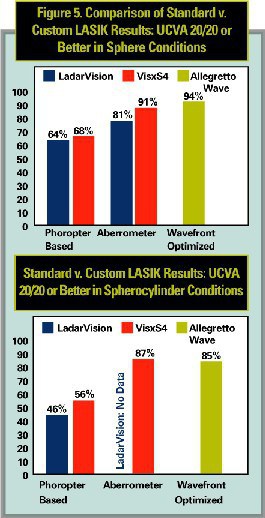
- Cause and effect. Presumably, wavefront treatments induce fewer aberrations than conventional treatments. But is that due to improvements in the lasers and overall ablation shapes, or to the use of the aberrometer? Aberrometer-based wavefront treatments show improved results over prior, phoropter-based, same-laser standard LASIK treatments. Figure 5 compares the outcomes for each laser with their own standard LASIK approvals from the past. However, no study proves that the aberrometer was responsible for the improvements. They may be due to technology upgrades in the laser and improved nomograms, rather than to the “custom” treatments.
Emphasizing this point, Figure 5 also shows that the Allegretto Wave’s wavefront-optimized, phoropter-based LASIK results outperformed the custom treatments. The fact that improvements in aberrations could not be shown in the FDA studies raises the question: what are we paying for?
The race to wavefront LASIK has led to better understanding of aberrations, visual function and laser technologies. LASIK results from the WaveLight Allegretto Wave FDA clinical trials show that wavefront-optimized profiles in treatments based on phoropter refractions outperform the custom results with the LadarVision and Visx S4 lasers, without the added costs of the aberrometer, higher click-fees, increased exam time and staff training.
When deciding which LASIK is best for our patients, surgeons will look at which systems yield the best results. Now that four systems are approved, I encourage surgeons to conduct their own analysis. In my analysis, wavefront-optimized, refraction-driven LASIK produced superior results to aberrometer-guided, customized LASIK.
Dr. Kezirian is a partner in SurgiVision Refractive Consultants, LLC, which runs the United States clinical trials for the WaveLight Allegretto Wave Excimer Laser System. He serves as a consultant to Lumenis Inc., the distributor of the Allegretto laser.
